Retinopathies and vascular lesions
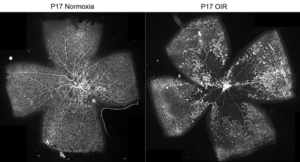
 Impairment of blood vessels in the retinal vasculature has been implicated in several human diseases, including retinopathy of prematurity and diabetic retinopathy. Retinal vessel in this pathological context show endothelial cell dysfunction, breakdown of the blood-retinal barrier, ischemia-induced retinal neovascularization, and expansion of the extracellular matrix, resulting in the outgrowth of fibrovascular tissue at the vitreoretinal interface. Defining the molecular pathways distinguishing pathological neovascularization from normal vessels is critical to controlling these blinding diseases. Our objectives aim to explore a new molecular player and signaling pathway, during ischemic retinopathy using mice model of retinopathy (Oxygen induced retinopathy OIR), transgenic approaches, and development of pharmacological tools.
Impairment of blood vessels in the retinal vasculature has been implicated in several human diseases, including retinopathy of prematurity and diabetic retinopathy. Retinal vessel in this pathological context show endothelial cell dysfunction, breakdown of the blood-retinal barrier, ischemia-induced retinal neovascularization, and expansion of the extracellular matrix, resulting in the outgrowth of fibrovascular tissue at the vitreoretinal interface. Defining the molecular pathways distinguishing pathological neovascularization from normal vessels is critical to controlling these blinding diseases. Our objectives aim to explore a new molecular player and signaling pathway, during ischemic retinopathy using mice model of retinopathy (Oxygen induced retinopathy OIR), transgenic approaches, and development of pharmacological tools.
 We have recently demonstrated that the Wnt/fzd pathway is via the canonical pathway (dependent on β-catenin) and the non-canonical pathway ( via the planar cell polarity) control vascular permeability and but also retinal angiogenesis.
We have recently demonstrated that the Wnt/fzd pathway is via the canonical pathway (dependent on β-catenin) and the non-canonical pathway ( via the planar cell polarity) control vascular permeability and but also retinal angiogenesis.
Our projects are actually focus on these pathways and its involvement in ischemic retinopathy. We have begun to have a proof of concept that the Wnt/fzd pathways could be efficient new target to treat aberrant angiogenesis and BRB impairment in ischemic retinopathies.
